Material Science and Engineering
Layered oxides for rechargeable zinc batteries
A technique of microwave synthesis of layered oxides enables high-capacity aqueous zinc-ion batteries.
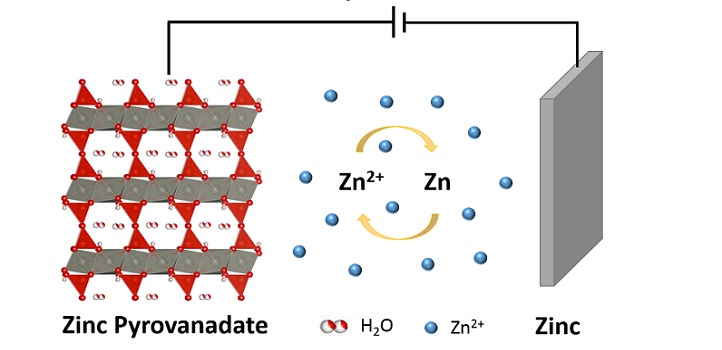
Layered oxides can form the basis of high-performance materials for battery electrodes. A KAUST team has developed a cheap and simple technique that creates this crucial element for rechargeable zinc-ion cells.
Lithium-ion batteries power most of our everyday electronic devices, such as cell phones and laptop computers. But there is a growing need to store energy on much larger scales, such as retaining the electricity generated by solar cells for use at night. Scaling lithium-ion battery technology up to such an industrial-level application is expensive and presents serious safety issues, including toxicity and the flammability of the electrolytes.
The team, led by Husam Alshareef, is instead developing zinc-ion batteries that use a water-based electrolyte, which has the advantages of being air stable, safe, environmentally friendly and cheap. “Aqueous batteries based on zinc ions can offer a safer, cost-effective solution to lithium ion batteries for grid storage,” says Alshareef. “Further, they use more environmentally friendly materials than lead-acid batteries.”
Lithium-ion and zinc-ion batteries work by electrically storing ions in an electrode. During charging, ions flow through an electrolyte from one electrode to another, where they are captured by a process known as intercalation. This means that electrode materials are key to optimizing a battery’s performance.
One family of materials that has shown much promise in recent zinc-ion battery research is vanadium-based compounds. These materials have a layered and very open atomic-crystal structure with plenty of spaces for trapping and storing zinc ions.
The team has now developed a microwave approach to rapidly synthesize ultralong zinc pyrovanadate (Zn3V2O7(OH)2·2H2O) cathodes. They mixed Zn(NO3)2·2H2O with commercial NH4VO3 powder, each dissolved in deionized water, and applied microwave radiation to induce a reaction. The resulting material was then dried before being used in a battery.
This zinc-ion battery, they show, can achieve an energy density of as much as 214 watt-hours per kilogram, which is much higher than previously reported aqueous zinc-ion batteries and commercial lead–acid batteries, with improved stability.
The team believes their microwave technique could also be useful for creating other metal pyrovanadate compounds, explains Chuan Xia, Ph.D. student and lead author of the study. “We have already made compounds in which zinc is replaced with other cations that create larger metal-oxide polyhedra that are able to intercalate even higher amounts of zinc ions”, says Xia.
References
- Xia, C., Guo, J., Lei, Y., Liang, H., Zhao, C. & Alshareef, H.N. Rechargeable aqueous zinc-ion battery based on porous framework zinc pyrovanadate intercalation cathode. Advanced Materials 29, 1705580 (2017).| article
You might also like

Chemistry
Turning infrared solar photons into hydrogen fuel

Applied Physics
A single additive enables long-life, high-voltage sodium batteries

Bioengineering
Smart patch detects allergies before symptoms strike

Applied Physics
Two-dimensional altermagnets could power waste heat recovery

Applied Physics
Interface engineering unlocks efficient, stable solar cells

Applied Physics
The right salt supercharges battery lifespan

Applied Physics
Light-powered ‘smart vision’ memories take a leap forward

Applied Physics