Bioscience
Single small-molecule model reveals insights into human embryo development
A unique embryo-like cellular model offers a scalable platform for studying human embryogenesis, fertility, and developmental disorders.
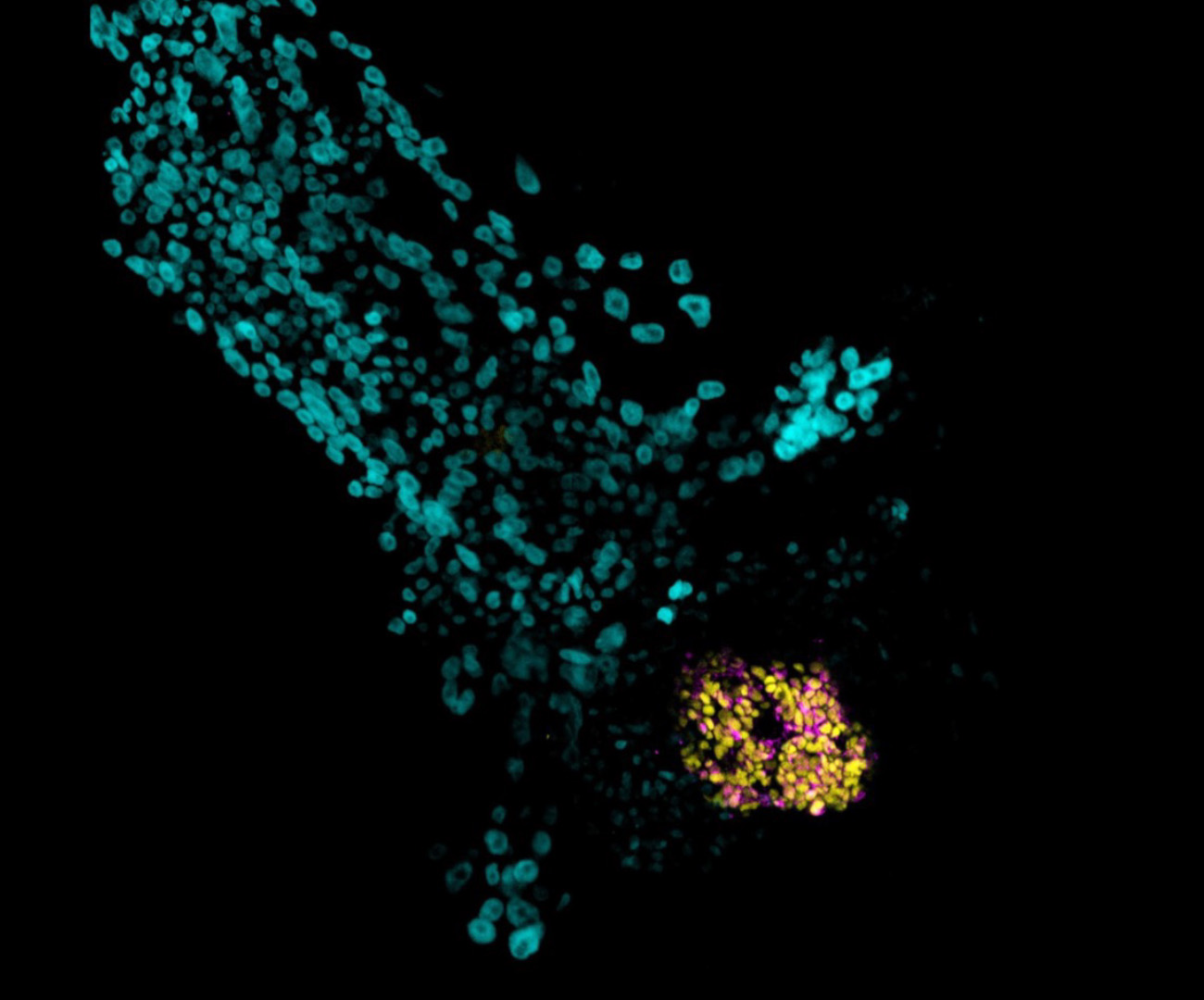
A simple yet efficient method of generating models of early human embryo development offers a robust platform for investigating infertility and developmental disorders. Developed by an international team led by KAUST, the model is already providing biological insights into the earliest stages of blastocyst formation[1].
“We were motivated by a fundamental question in biology: how does a single cell organize into a complex embryo during the earliest stages of development?” says Mo Li, who supervised the study. “These stages, particularly blastocyst formation, are critical for implantation and successful pregnancy, yet they remain poorly understood.”
Amid global fertility decline and increasing reliance on treatments such as in vitro fertilization (IVF), there is a clear need to develop reproducible, ethical models of blastoids – early embryo-like structures – from human naïve pluripotent stem cells.
“Many IVF failures occur at or before the blastocyst stage, highlighting the need to better understand early embryonic mechanisms and improve clinical outcomes,” says Arun Pandian Chandrasekaran, a postdoc who worked on the project.
A fortuitous discovery
The team made an unexpected discovery in the lab while they were exploring potential new models for trophectoderm, one of the key lineages of the blastocyst. They observed an unusual developmental phenotype during experiments, and traced the trigger for this phenotype to a specific small molecule.
“The reagent dimethyl sulfoxide (DMSO) is widely used as a solvent to dissolve a broad range of chemicals and as a cryoprotectant cell preservation. We noticed that it appeared to be doing something far more profound in our setup, so we followed the science,” says Li.
“Existing trophectoderm and blastoid models typically rely on combinations of multiple signaling molecules,” says Samhan Alsolami, a former KAUST team member, now at the Salk Institute for Biological Studies in California, United States. Alsolami remains affiliated with KAUST via the Ibn Rushd fellowship program. “We found that DMSO alone could induce trophectoderm-like differentiation. Building on this, we showed that DMSO is sufficient to drive robust blastoid formation from human pluripotent stem cells without the need for any additional factors,” he explains.
The simplicity of the model surprised the team, who were expecting to need a complex cocktail of molecules and signals to generate functioning blastoids.

A first for embryogenesis research
Using this model, the researchers uncovered a crucial biological mechanism that helps the embryo correctly form its first cavity. They found that tiny structures called lysosomes, located inside cells, must function properly for this cavity to form. The team proved that the molecular pump V-ATPase plays a central role in driving and regulating blastoid cavitation. Furthermore, DMSO treatment upregulates V-ATPase activity to ensure the cavity forms correctly.
“This discovery opens the door to numerous further investigations, including examining why early developmental failure happens in some embryos but not others,” says Yiqing Jin from the KAUST team. “Our model may help researchers understand early implantation-related defects, pregnancy loss, and developmental abnormalities that begin before pregnancy is even clinically recognized.”
“The model could also be used to investigate congenital disease mechanisms in a controlled setting,” adds Alsolami. “The system’s simplicity and reproducibility make it well suited for mechanistic studies and screening approaches.”
Future goals
The KAUST team is extending this work to build models that capture all stages of embryogenesis with greater fidelity. In particular, Chandrasekaran is developing a model that captures the features of day 14 of human embryogenesis.
“Our vision is not only to generate embryo models but to use them as discovery platforms,” concludes Li. “We hope to combine stem cell biology with imaging, genomics, and functional perturbation approaches to answer deeper questions about how mammalian embryos self-organize, why development sometimes fails, and how these earliest events influence health later in life.”
Reference
- Alsolami, S., Chandrasekaran, A.P., Jin, Y., Wang, Y., Zhang, L., Shakir, I.M., Zhang, Y., Siddique, A., Ramos-Mandujano, G., Yuan, B., Ayach, M., Seara-Vila, A., Fan, Z., Fu, S., Zhang, H., Xin, S., AlDakhil, K.K., Belmonte, J.C.I., Zhang, J., Yu, Y., & Li, M. A single small molecule-based human embryo model reveals a requirement for V-ATPase in mammalian blastocyst cavitation. Cell Research (2026) | article
You might also like

Bioscience
Can AI finally bring order to biology’s data deluge?

Bioengineering
Bio-inspired network structures for next-generation AI

Bioscience
Robust workflow built for chemical genomic screening

Bioscience
Cell atlas offers clues to how childhood leukemia takes hold

Bioscience
Hidden flexibility in plant communication revealed

Bioscience
Harnessing the unintended epigenetic side effects of genome editing

Bioscience
Mica enables simpler, sharper, and deeper single-particle tracking

Bioengineering



