Chemistry
Branching out for the right fuels
Metal-organic frameworks are effective in separating linear from branched paraffins.
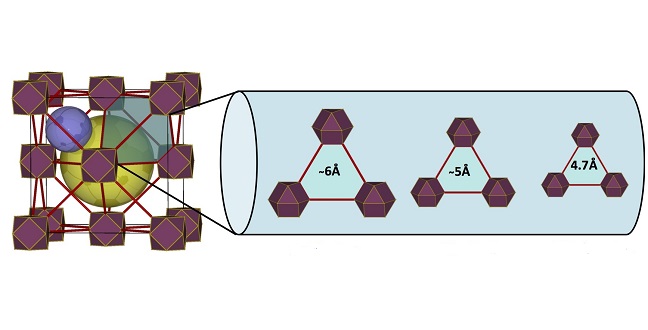
The porous structure of MOFs (left) allows for a molecular exclusion of branched paraffins versus linear ones by tuning the MOF pore structure to the right size.
Reproduced with permission from reference 1© 2016 John Wiley & Sons.
Porous chemical compounds that can efficiently separate paraffins used in oil and gasoline refinement have been developed by researchers at KAUST. The porous structure of the materials can be precisely tuned at the atomic level, enabling a fully designed molecular selection process1.
Refinement remains the key process to produce fuels from crude oil. However, achieving high-octane fuels is costly and energy-intensive; their chemical similarity means that it is difficult to separate the components for high and low-quality gasoline or diesel fuels. Present methods utilize the different boiling points and evaporation behavior of the relevant chemical molecules.
One alternative is to separate molecules according to their structural features, size and shape. For example, in high-octane gasoline, it is better to have paraffins that have a branched rather than a straight, linear structure. Filtering these different molecules can be achieved by using highly porous materials, and in particular metal-organic frameworks (MOFs).
MOFs consist of inorganic building blocks linked together into a network via organic molecules, forming extended two or three-dimensional structures. The periodic array of organic and inorganic building blocks creates large empty spaces, and these pores can be occupied by guest molecules of the right size.
One attractive feature of MOFs is that the size of the open pores can be tuned precisely by using different organic linker molecules. This creates versatile systems, said Mohamed Eddaoudi, who led the research team.
“Using the molecular building block approach, it is possible to deliberately construct/assemble a MOF with predetermined triangular window sizes that enable easy passing of certain molecules and prevent passing of relatively larger molecules. In this case, the passing of normal paraffin and blocking of branched ones,” Eddaoudi noted.
In this case, the size of the triangular window, the sole entrance to the pores of the reported fcu-MOF, varied from about 4.7 Angstrom (about half a nanometer) to about 6 Angstrom, with the 4.7 Angstrom window size being ideal to separate linear from branched paraffins. Indeed, in adsorption experiments, the linear paraffins were highly adsorbed by the MOF, whereas branched ones were not. The separation takes place at room temperature, highlighting the practical potential of this approach.
While at present the selection process is based on adsorption/molecular exclusion process of the target molecules in the MOFs, the team is working to develop ultrathin MOFs that could separate the paraffins by operating like a sieving membrane.
These membranes could also be used for other key energy-intensive separations, added Eddaoudi. “We are aiming to use the same strategy based on MOFs to target other very difficult separation such as olefin/paraffin, oxygen/nitrogen, or nitrogen/methane,” he said.
References
- Assen, A.H.,Belmabkhout, Y., Adil, K., Bhatt, P.M., Xue, D.-X., Jiang, H. & Eddaoudi, M. Ultra-tuning of the rare-earth fcu-MOF aperture size for selective molecular exclusion of branched paraffins. Angewandte Chemie International Edition 54, 14353 –14358 (2015). | article
You might also like

Chemical Engineering
Natural solvents enable fully bio-based membranes

Chemistry
Turning infrared solar photons into hydrogen fuel

Applied Physics
Natural polymer boosts solar cells

Chemistry
Disruptive smart materials flex with real world potential

Chemistry
Catalysts provide the right pathway to green energy

Chemistry
Hollow molecules offer sustainable hydrocarbon separation

Chemistry
Maximizing methane

Chemistry



