Chemical Engineering
Separation a sweet success
Cone-shaped sugar structures can be connected together to form selectively porous nanofiltration membranes.
Sugars can make excellent gatekeepers for separating mixtures of molecules. A KAUST team has discovered a way to make a selectively porous membrane by linking large numbers of sugar structures, known as cyclodextrins, to form a thin film.
Until now, membranes made from disordered tangles of long-chain polymers have been the “workhorse materials” of industrial-scale small-molecule separation, says Klaus-Viktor Peinemann, who led the team at KAUST. Molecules cross these membranes by slipping through gaps between the interlinked polymer chains. But, because the gaps vary in size, the membranes are not perfectly selective in the molecules they allow to cross.

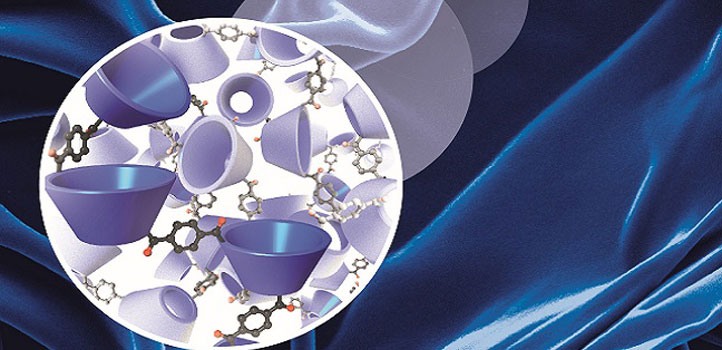
The cyclodextrins (blue cones, inset image) linked together to form a wrinkled, selectively porous membrane (main image).
© Reproduced with permission from reference 1. © 2017 WILEY‐VCH Verlag GmbH & Co.
Cyclodextrins are funnel-shaped sugar structures in which the shape and size of the cavity controls which molecules can pass through it. With fixed-shape pores, cyclodextrins are attractive alternative building blocks for selectively permeable membranes, says Luis Francisco Villalobos, first author of the paper and Ph.D. student in Peinemann’s lab. The hurdle was that nobody had found a way to connect cyclodextrins to form a membrane.
The team used a “very old strategy” to overcome this problem, according to Villalobos, adapting the synthetic approach already used by industry to make polyamide membranes for reverse osmosis separations, such as water desalination. Compared to the building blocks used to make polyamide membranes, “cyclodextrins are bulkier, less reactive, less soluble and tend to aggregate in aqueous solutions,” Villalobos adds.
To get them to react, the key was to find the right pH.
Under strongly alkaline conditions, the cyclodextrins lose a proton, forming an ionic structure with boosted reactivity and solubility. The team exploited this discovery to create continuous films of cyclodextrins, which proved to have excellent porosity. “The membranes we prepared allowed solvents to permeate more than ten times faster than equivalent commercially available membranes,” Villalobos says.
“Moreover, they can discriminate between molecules based on their shape thanks to the cavities of the cyclodextrins,” Villalobos adds. As a proof-of-principle, the team separated a mixture of two molecules of similar size but different shape.
Because cyclodextrins are a cheap and readily available material produced from starch, these membrane could be a very attractive material for separating mixtures of molecules on an industrial scale, Peinemann says.
“But this is just the first step—cyclodextrins are only one example of an intrinsically porous building block,” Villalobos adds. “Similar strategies could be followed to fabricate films using other building blocks with cavity sizes that are attractive for targeted, industrially relevant separations.”
References
-
Villalobos, L.F., Huang, T. & Peinemann, K.V. Cyclodextrin films with fast solvent transport and shape-selective permeability. Advanced Materials 29, 160664 (2017).| article
You might also like

Chemical Engineering
Engineered membranes made from corn proteins

Chemical Engineering
Clean jet fuel chemistry takes flight

Chemical Engineering
Natural solvents enable fully bio-based membranes

Chemical Engineering
Urban air pollution goes up in smoke

Chemical Engineering
Rethinking machine learning for frontier science

Chemical Engineering
Magnetic nanoparticles capture microplastics from water

Chemical Engineering
Biogas upgrading goes with a swing

Chemical Engineering




