Mechanical Engineering
Antibubbles make a splash
Stable air layers that wrap around liquid drops can retain their remarkable geometries a thousand times longer than theory predicts.
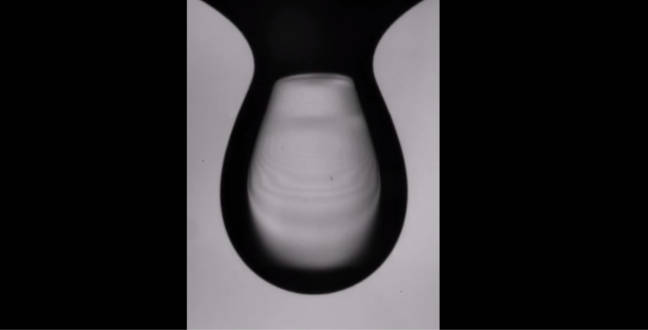
When a viscous oil droplet enters into another liquid, an air cushion wraps around its long tail and eventually breaks off – creating free-floating cylinders of air with surprisingly long lifetimes..
© KAUST 2015
Natural processes, including cloud formation and soil erosion, and technology such as inkjet printers are based on interactions between droplets and surfaces. One area of investigation for climate scientists and engineers is low velocity impacts — for example, secondary splashes that occur after the primary contact, or drops ejected from a boiling interface. These types of collisions are hard to predict, as they involve complex physical interactions.
KAUST Research Scientist Erqiang Li and Professor of Mechanical Engineering Sigurdur Thoroddsen from the University’s Physical Science and Engineering Division, along with collaborators in France and the Netherlands, used advanced video cameras to study drop impacts between different liquids.
At low velocities, drops tend to either bounce away or enter into target liquids on a thin, hemispherical air cushion that eventually ruptures into microbubbles. The team realized that they might be able to control the geometry of these air sheets if they used droplets that are more viscous.
To experiment, the researchers placed a syringe filled with thick, viscous silicone oil a few millimeters above a pool containing lower-viscosity oil. By gradually increasing droplet viscosity, air sheets began to envelop the heavy liquids as they fell into the light oil. After analyzing several thousand video clips, the team mapped out the parameters needed to produce seldom-seen spherical antibubbles on demand.
At the upper limits of viscosity, the droplets stay connected to the syringe nozzle along long, thin threads of oil during the initial impact. When the droplet and its long tail enter into the pool, air sheets also wrap around these structures and produce cylindrical films of air that are stable for thousands of times longer than expected.
When the team used laser-based interferometry to scrutinize the cylinders, they were surprised to see the films can become narrower than 100 nanometers before breaking. At these scales, stability theory predicts their disintegration in a few microseconds through the same mechanism that breaks up the water stream running out of faucets.
“Soap molecules could stabilize the layers, but there are no surfactants involved here,” said Thoroddsen. “Our results suggest the stabilization force is the viscous lubrication forces within the submicron air layer. This shines new light on thin film instability and also introduces many more questions.”
References
- Beilharz, D., Guyon, A., Li, E. Q., Thoraval, M.-J. & Thoroddsen, S. T. Antibubbles and fine cylindrical sheets of air. Journal of Fluid Mechanics 779, 87–115 (2015). | article
You might also like

Mechanical Engineering
Green hydrogen from fluctuating power sources

Mechanical Engineering
Cracking clean fuel combustion

Mechanical Engineering
Tiny sensor could transform head injury detection

Mechanical Engineering
Electrocatalytic CO2 upcycling excels under pressure

Chemical Engineering
Rethinking machine learning for frontier science

Mechanical Engineering
Falling water forms beautiful fluted films

Mechanical Engineering
Innovative strain sensor design enables extreme sensitivity

Mechanical Engineering



