Bioscience
Doubled up protein protects against cancer
Insight into additional functions of a key cancer protein could pave the way for new anti-cancer drugs.
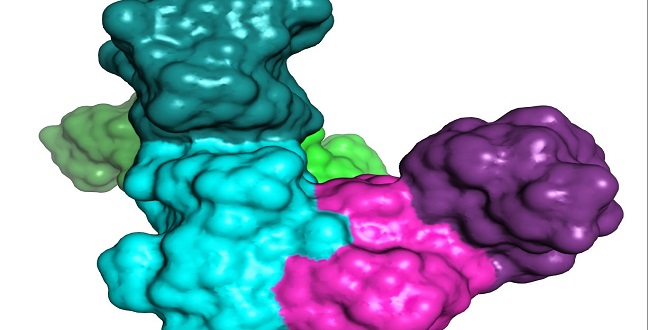
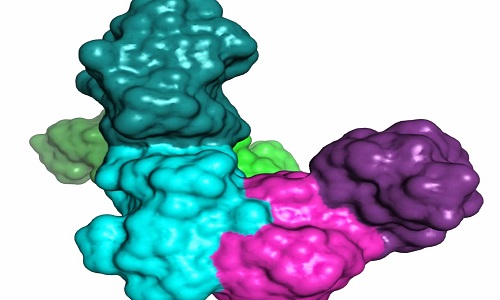
The theoretical model of the complex between a p85 dimer and PTEN.
© 2015 KAUST
A protein that is faulty in many cancers has a previously undiscovered function, reveals work by researchers from KAUST and MD Anderson Cancer Center (Houston, Texas, U.S.). The findings provide insight into molecular mechanisms of tumor development and could lead to new treatments.
The research focused on the PI3 kinase pathway that regulates many cellular processes. PI3 kinases are enzymes comprised of two subunits: p110, the catalytically active subunit, and p85, which binds to p110 and ensures it is activated only by specific stimuli. Mutations in p85 prevent it from controlling p110, leading to unregulated PI3 kinase activity, and are often found in patients with cancer.
Previous research has shown that p85 can bind to other p85 molecules to form dimers. p85 can also bind to a protein called PTEN, which protects against cancer by opposing the PI3 kinase pathway. However, the significance of these interactions has been unclear.
“We wanted to understand in molecular detail the functional outcome of p85 dimerization and its interaction with PTEN,” explains Stefan Arold, a senior investigator in the study and an Associate Professor in KAUST’s Division of Biological and Environmental Science and Engineering.
The researchers first identified the regions of p85 that associate during dimerization, before making mutant p85 that was unable to dimerize. They found that the mutant p85 could not bind to PTEN in cultured cancer cells, but it could still interact with p110. They went on to show that the binding of p85 dimers protects PTEN from degradation, ensuring that cellular levels of PTEN are sufficient to effectively counteract PI3 kinase activity.
“Our findings reveal a previously unknown function of the p85 dimer and identify the mechanism by which an equilibrium of p85 monomers and dimers balances the opposing activities of PI3 kinase and PTEN,” explains Arold. “Disrupting this equilibrium could lead to cancer by enhancing activation of the PI3 kinase pathway.”
The researchers found evidence for the proposed mechanism by looking at the genetics of patients with different types of cancer. Many of the patients had mutations that prevent the dimerization of p85 or the interaction of p85 dimers with PTEN.
“Molecules that increase the number of p85 dimers and their interaction with PTEN might be viable therapeutic agents against cancer that is caused by PI3 kinase pathway hyperstimulation,” says Arold. “We will now try to obtain atomic-resolution images of the interaction between p85 and PTEN to provide a stepping-stone for the rational development of drugs that target this process.”
References
- Cheung, L.W.T., Walkiewicz, K.W., Besong, T.M.D., Guo, H., Hawke, D. H., Arold, S.T. & Mills, G.B. Regulation of the PI3K pathway through a p85α monomer–homodimer equilibrium. Elife 4, e06866 (2015).| article
You might also like

Bioscience
Robust workflow built for chemical genomic screening

Bioscience
Cell atlas offers clues to how childhood leukemia takes hold

Bioscience
Hidden flexibility in plant communication revealed

Bioscience
Harnessing the unintended epigenetic side effects of genome editing

Bioscience
Mica enables simpler, sharper, and deeper single-particle tracking

Bioengineering
Cancer’s hidden sugar code opens diagnostic opportunities
Bioscience
AI speeds up human embryo model research
Bioscience



