Material Science and Engineering
Building better blockers for solar cells
An oxide layer optimized to selectively transmit electrons can improve the efficiency of organic solar cells beyond 10 percent.
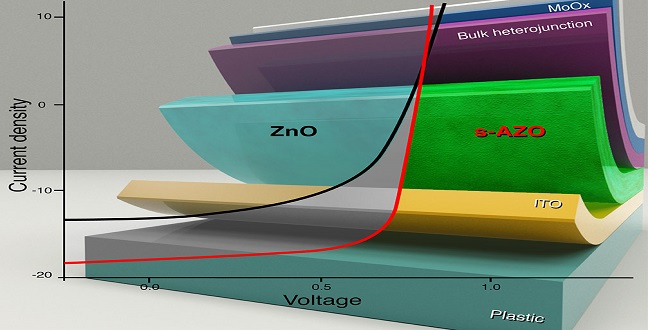
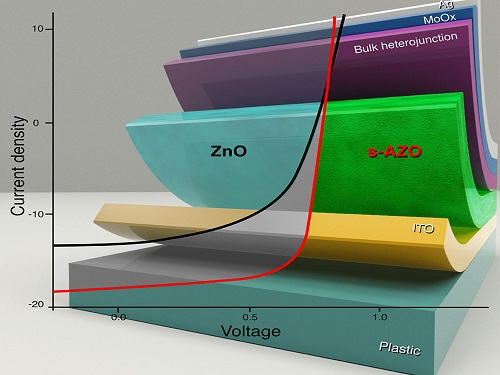
The operation characteristics of organic thin-film solar cell devices can be improved with the introduction of a zinc oxide layer.
© 2015 KAUST
An ingenious method to improve the efficiency of polymer solar cells has been developed by a team of researchers at KAUST.
There is a widespread push to use organic materials in the conversion of sunlight to electricity. These substances are advantageous over silicon in terms of cost, production at relatively low temperatures, and compatibility with flexible plastic substrates.
When a polymer absorbs light, the optical energy creates an electron and its positively charged counterpart known as a hole. A current flows through the device when the electron moves one way and the hole go the other. To enable this, polymer solar cells comprise many different layers. These can include the light-absorbing active polymer layer, a hole collecting/electron blocking layer, and an electron collecting/hole blocking layer.
“In thin film solar cells, selective contacts are crucial to making sure that electrons go only to the cathode and holes go to the anode,” says Aram Amassian, assistant professor at the KAUST Solar Center. “While significant effort has been put into improving the photoactive light harvesting material, there has been far less attention on the selective contacts. Poor contacts can mean a solar cell with supposed 10 percent efficiency behaves instead like a 5-6 percent device, if not worse.”
Amassian and colleagues have now shown that an electron-collecting layer made with zinc oxide layer doped with aluminum and nitrogen atoms can improve the power conversion efficiency of solar cells made from a wide range of organic materials.
Lethy Jagadamma, the study’s lead author, created a series of solar cells from polymer-fullerene blends, using a zinc oxide layer, with or without aluminum and/or nitrogen doping. The team also tried several methods for creating the zinc-oxide layer: thermal evaporation or solution processing using either nanoparticles or an amorphous sol-gel.
A comparison of the operative characteristics of each device indicated that the solution processed solar cells were best, ultimately achieving an efficiency of 10.2 percent on glass and 8.2 percent on plastic substrates. The team also found that they could improve the efficiency further by introducing a small amount of ammonia to the solution when processing the devices. The ammonia reduced the roughness of the oxide layer, made it more compact and further doped it with nitrogen.
“We are confident that further improvements in interfacial science and engineering of contacts will lead to even greater breakthroughs across emerging photovoltaic technologies,” says Amassian.
References
- Jagadamma, L. K., Al-Senani, M., El-Labban, A., Gereige, I., Ngongang Ndjawa, G. O. et al. Polymer solar cells with efficiency >10% enabled via facile solution-process Al-doped ZnO electron transporting layer. Advanced Energy Materials 5, 1500204 (2015).| article
You might also like

Chemistry
Turning infrared solar photons into hydrogen fuel

Applied Physics
A single additive enables long-life, high-voltage sodium batteries

Bioengineering
Smart patch detects allergies before symptoms strike

Applied Physics
Two-dimensional altermagnets could power waste heat recovery

Applied Physics
Interface engineering unlocks efficient, stable solar cells

Applied Physics
The right salt supercharges battery lifespan

Applied Physics
Light-powered ‘smart vision’ memories take a leap forward

Applied Physics



