Chemical Engineering
A one way trip for water
A material for filtration that is easy and cheap to produce could aid water treatment, solvent filtration and membrane-catalysis.
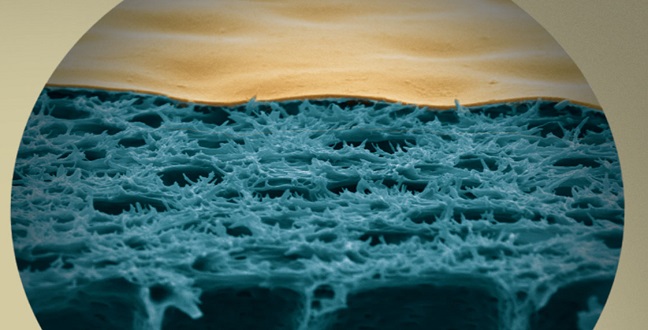
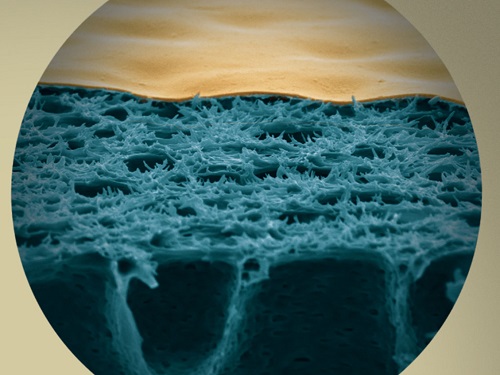
The process of complexation-induced phase separation produces membranes with a dense surface layer (gold-colored) on a more porous region (blue).
© 2015 KAUST
A cheap and cost-effective technique for making water- and solvent-filtering polymers has been developed by a team of researchers at the King Abdullah University of Science and Technology (KAUST)1.
Membranes are thin films permeable by some particles but a barrier to others and are used for removing salt or contaminants such as dirt, bacteria or even viruses from water. A common method for constructing such structures is to combine the thin but dense membrane, or skin, with a thicker porous material that provides mechanical support.
The processes used to construct these composite membranes are complicated or require expensive materials, which prevents production on a commercial scale.
Klaus-Viktor Peinemann and his PhD student Luis Francisco Villalobos from the KAUST Physical Science and Engineering Division have come up with a simple and scalable process for building multi functional membranes. Their composite membrane needs only one casting step and one polymer solution to prepare and is then treated with two successive baths, as Peinemann explains. “This process is simple and we can easily prepare defect-free coatings with thicknesses that are difficult to obtain with conventional coating procedures.”
The KAUST team named their technique ‘complexation-induced phase separation’. It works by connecting long chains of polymer molecules — referred to as cross-linking — with metal ions. Peinemann and his colleagues made a solution comprised of a polymer and an organic solvent, deposited it on a substrate and dipped it in a bath containing the metal ions dissolved in the same solvent.
This was followed by immersion in a second metal-free non-solvent bath. They could show that complexation-induced phase separation produces a dense surface layer of cross-linked polymer–metal complexes separated by a well-defined interface from a more porous region beneath it (see image).
The advantages of the new phase separation process are that it is fast and easy to control, which enabled the researchers to tailor the precise structure of their asymmetric membrane. For example, they were able to vary the thickness of the skin between fifteen nanometers and six micrometers. They could also alter the morphology of the membrane surface and the porosity of the support structure by altering the type of metal ion used, which included silver, palladium, cobalt, nickel and copper.
“So far we have demonstrated the complexation-induced phase separation process with two polymers,” says Peinemann. “Next we hope to identify a number of other polymers suitable for this new formation process.”
References
- Villalobos, L. F., Karunakaran, M. & Peinemann, K.-V. Complexation-induced phase separation: Preparation of composite membranes with a nanometer-thin dense skin loaded with metal ions. Nano Letters 54, 3166–3171 (2015). | article
You might also like

Chemical Engineering
Urban air pollution goes up in smoke

Chemical Engineering
Rethinking machine learning for frontier science

Chemical Engineering
Magnetic nanoparticles capture microplastics from water

Chemical Engineering
Biogas upgrading goes with a swing

Chemical Engineering
Stronger, lighter, cheaper: a new route to carbon fiber production

Chemical Engineering
Unveiling the role of biomass-burning aerosols in atmospheric reactions

Chemical Engineering
Slashing industrial emissions using a hybrid model approach

Chemical Engineering



