Electrical Engineering
A radical solution comes from mixing tools
The molten surface of a sodium-based material could assist the direct conversion of methane to useful building blocks.
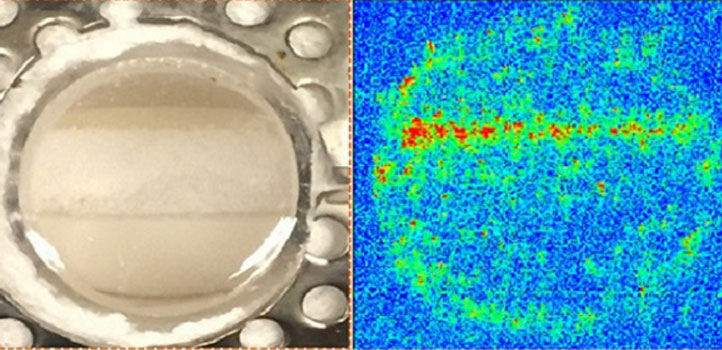
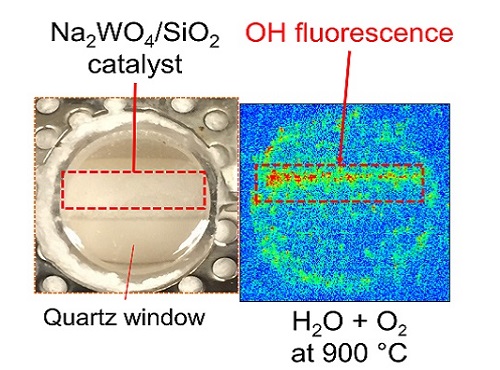
The active species in the catalytic reaction that exits on the molten surface of the sodium tungstate is sodium peroxide.
Reproduced with permission from reference 1 © 2017 Wiley-VCH Verlag GmbH & Co.
For natural gas to be efficiently converted into useful industrial products requires the right catalytic process. Researchers from KAUST and the US combine state-of-the-art techniques for material characterization to demonstrate a unique reaction pathway that shows that molten catalysts based on sodium can provide all the chemical species required to optimize the process.
Free radicals, molecules with an unpaired valence electron, such as the hydroxyl radical, play a crucial role in the industrially important conversion of natural gas, primarily methane, to ethylene: a vital organic compound that forms the building blocks of many commodities and polymers. To enhance this process, known as oxidative coupling, it is vital to develop selective catalysts.
The KAUST team—led by Kazuhiro Takanabe and his student Abdulaziz Khan—used in situ tools to measure the state of a catalyst under reaction conditions. They discovered that the active species in the catalytic reaction that exits on the molten surface of the sodium tungstate, a chemical necessary for the reaction, is sodium peroxide. This catalyst is unique in that instead of activating methane directly, it firsts activates water and then generates gaseous hydroxyl radicals.
Oxidative coupling of methane converts methane and oxygen to ethylene in a single reactor. Previous research in Takanabe’s lab had indicated that by using sodium tungstate at temperatures above 700 °C, the presence of water can both the increase the rate of methane conversion and enhance product selectivity. This could potentially occur via the formation of hydroxyl radicals and sodium peroxide, but there was no direct evidence for the presence of these species.
Now, Takanabe and his co-authors provide direct evidence for the formation of these free radicals on the molten surface of sodium tungstate. They combine a wide range of experimental techniques, including X-ray diffraction, scanning transmission electron microscopy, laser-induced fluorescence spectrometry and X-ray photoelectron spectroscopy, to observe an outer layer of molten sodium tungstate that is rich in sodium hydroxide. “We exclusively identified the catalyst’s active phase in a unique state under reaction conditions,” explains Takanabe.
This in turn confirms that a sodium-based catalyst can form hydroxyl radicals from a mixture of oxygen and water, a reaction that has never been seen. “This catalyst and the unique reaction pathway have great potential for use in various catalytic reactions for natural gas conversion, petroleum refinery and combustion reactions,” says Takanabe.
More broadly, this success also demonstrates the importance of combining in situ spectroscopic and microscopic techniques to better understand high-temperature gas-phase chemistry.
References
-
Takanabe, K., Khan, A. M., Tang, Y., Nguyen, L., Ziani, A., Jacobs, B.J., Elbaz, A.M., Sarathy, S.M. & Tao, F. Integrated in situ characterization of molten salt catalyst surface: Evidence of sodium peroxide and OH radical formation. Angewandte Chemie 56, 10403-10407 (2017).| article
You might also like

Electrical Engineering
Quantum dot lasers outshine expectations

Bioengineering
Self-aware biosensors boost digital health monitoring

Bioengineering
Smart patch detects allergies before symptoms strike

Computer Science
Green quantum computing takes to the skies

Electrical Engineering
Micro-LEDs boost random number generation

Bioengineering
Sensing stress to keep plants safe

Computer Science
Sweat-sniffing sensor could make workouts smarter

Electrical Engineering



