Electrical Engineering
Splitting water for the cost of a nickel
A simple treatment of nickel to make it an efficient catalyst for water splitting.
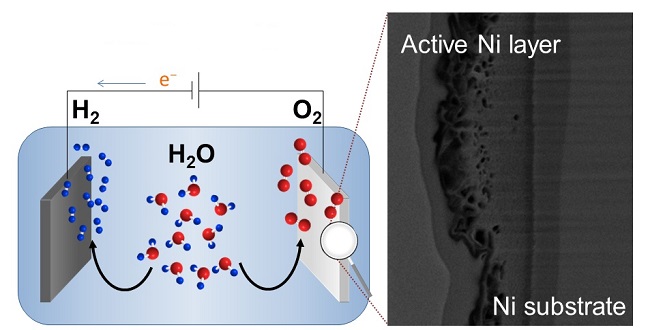
An electron microscope image of a nickel anode (right) that boosts the oxygen-evolution reaction that is important for water splitting (left).
© KAUST
A technique to create a material for cost-effective water electrolysis uses a simple chemical method for preparing nickel-based anodes to improve the oxygen-evolution reaction. Efficiency gains like this one developed by KAUST are important in evolving renewable energy.
Hydrogen stores a tremendous amount of energy and so offers great potential as a sustainable, carbon-free source of power. Hydrogen is also abundant on Earth, although predominantly in the form of water. Electrocatalysis can separate the hydrogen atoms from the oxygen atoms, but a crucial consideration is a process known as oxygen evolution. The rate of oxygen creation is known to affect the overall production rate of hydrogen; thus, scientists are searching for a catalyst to enhance this reaction.
Noble metals, such as iridium and ruthenium, have excellent oxygen-evolution performance but are very expensive. As a cheaper alternative, Ph.D. student Tatsuya Shinagawa and internship student Marcus Ng were guided by Kazuhiro Takanabe from the KAUST Catalysis Center to use a simple chemical method to improve the oxygen-evolution reaction.
Nickel-based electrocatalysts have shown encouraging performance in previous experiments. Notable amongst these is nickel-iron oxide; however, the cost and complexity of its synthesis is a drawback.
“Existing electrolyzers function in extremely alkaline or acidic environments, and these harsh conditions will be costly when driven by renewable energy sources,” says Shinagawa. “Also important is that most studies on electrocatalytic water splitting have been performed at room temperature, while higher temperatures are required in practical systems.”
The team’s protocol involves repeated redox cycling of nickel in a carbonate or phosphate electrolyte at moderate pH and temperature of approximately 55°C and the evaluation of the water-splitting process performance at elevated temperatures (up to 80°C)
Imaging the resultant nickel electrode with a scanning electron microscope indicated that the fabrication process restructures the surface to create a layer of nickel-oxide hydroxide more than one-millimeter thick. The 3D structure of this layer can trap weakly bound alkali-metal cations and water.
The team showed that their electrode exhibited greatly superior oxygen-evolution reaction performance compared to nickel-iron oxide under near-neutral pH conditions and at temperatures commonly used in industrial processing.
“We hope to follow this study by building a further understanding of the material’s properties and its performance,” says Shinagawa. “For example, an in-depth study on kinetics will identify the limiting reaction step over the prepared nickel-based electrocatalyst, which will lead to further improvement of the catalytic activity.”
References
-
Shinagawa, T., Ng, M. T.-K. & Takanabe, K. Boosting the performance of the nickel anode in the oxygen evolution reaction by simple electrochemical activation. Angewandte Chemie 56, 5061(2017).| article
You might also like

Computer Science
AI-based hydrogen plant models improve power grid stability

Electrical Engineering
Quantum dot lasers outshine expectations

Bioengineering
Self-aware biosensors boost digital health monitoring

Bioengineering
Smart patch detects allergies before symptoms strike

Computer Science
Green quantum computing takes to the skies

Electrical Engineering
Micro-LEDs boost random number generation

Bioengineering
Sensing stress to keep plants safe

Computer Science



