Plant Science
Glowing report on plant hormone function
A biological sensor can identify and quantify the activity of a little-known class of plant hormones, strigolactones.


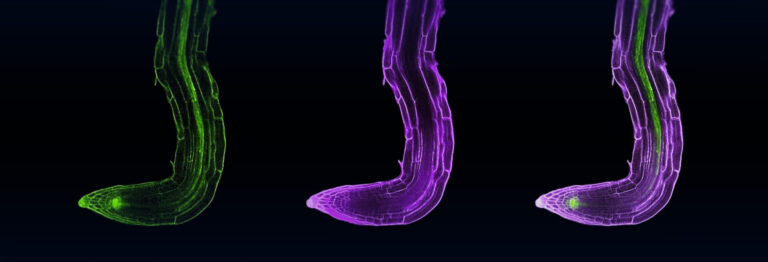
The new strigolactone sensor comprises a version of SMXL coupled to a yellow luminescent enzyme, luciferase, derived from fireflies.
© Shutterstock
Strigolactones are an important and diverse class of plant hormones. Now, an international team led by KAUST Associate Professor Salim Al-Babili and Professor Matias Zurbriggen from the University of Dusseldorf has developed a strigolactone sensor that can be genetically encoded into plant cells to help our understanding of plant development1.
Strigolactones regulate development within plants and facilitate communication with other organisms, such as parasites and symbiotic fungi. However, little is known about how these hormones work because there are few tools to investigate them.
According to Al-Babili, “Strigolactones are unstable and occur at very low concentrations, which makes these hormones difficult to study.” We don’t know the functions of the different strigolactones or how they trigger responses. What is known is that strigolactones are sensed in plant cells when they bind to a receptor protein known as D14, forming a complex.
This binding recruits another protein to the complex, SMXL, which is then degraded, activating downstream responses. The team harnessed this degradation process to develop their innovative sensor.
The new strigolactone sensor—which is genetically encoded so that it can be produced within living cells—comprises a version of SMXL coupled to a yellow luminescent enzyme, luciferase, derived from fireflies. When SMXL degrades, the luciferase is also degraded, resulting in reduced yellow luminescence.
To enable quantification of the effect of strigolactones, the sensor also incorporates a green luciferase derived from the sea pansy, an animal related to jellyfish and corals. The green luciferase is produced in the same quantities as SMXL but is not directly attached to it and is therefore not degraded when strigolactones are present. The ratio of the two colors of luminescence gives a very precise measure of the level of strigolactone activity.
The research team tested the functionality, sensitivity and specificity of their biosensor, and they have already used it to show that one form of strigolactones, known as the 2’R form, is more active in plants than the alternative 2’L form.
Thanks to its modular construction, the new tool can easily be modified—for instance by incorporating different SMXLs—to study other aspects of strigolactone signaling.
Al-Babili believes applications of the sensor include not just strigolactone signaling, but strigolactone synthesis and complex related regulatory networks in plants. Understanding these networks may facilitate future manipulation of plant development including, he says, “optimizing crop architecture, nutrient uptake and growth performance. The knowledge obtained could also be very useful for combating damaging parasitic weeds that rely on strigolactones to identify their hosts.”
References
- Samodelov, S.L., Beyer, H.M., Guo, X., … Al-Babili, S. et al. StrigoQuant: A genetically encoded biosensor for quantifying strigolactone activity and specificity. Science Advances 2,e1601266 (2016).| article
You might also like

Plant Science
Coevolution of the microbiome with its host plant supports environmental adaptation

Bioscience
Hidden flexibility in plant communication revealed

Plant Science
Reference genomes for rice’s wild relatives may boost future crops
Bioscience
Digging into the world of plant-growth-promoting microbes

Environmental Science and Engineering
Hydrogen storage solution could lie in lakes

Bioscience
Unraveling modern bread wheat from the genes up

Bioscience
Why do some plants thrive in saline conditions?
Bioengineering



