Electrical Engineering
Targeting cancer cells with tiny magnetic wires
Magnetic nanowires with weak magnetic fields and low frequencies can destroy cancer cells without generating heat.
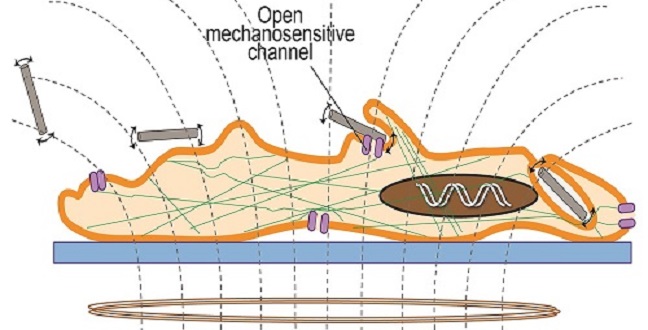
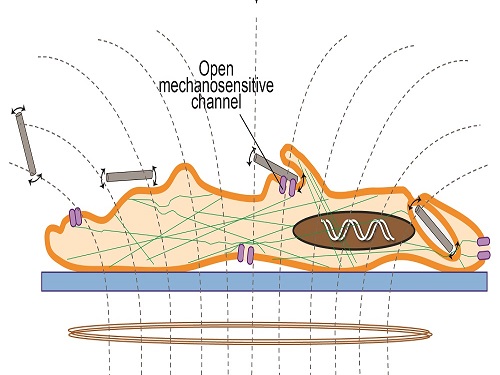
Applying the magnetic field triggered vibrations in the nanowires and generated changes in cell functions and structures that eventually led to cell death.
© 2015 KAUST
Incubating cancer cells with nickel nanowires before exposing them to a weak magnetic field can kill them, report researchers from KAUST1.
The multidisciplinary team— led by Jürgen Kosel from the Computer, Electrical and Mathematical Science and Engineering Division and Tim Ravasi from the Division of Biological and Environmental Science and Engineering — exposed colon cancer cells to various alternating magnetic fields. In early tests only a small percentage of the cells were destroyed. This indicated that the fields on their own were fairly harmless.
Colon cancer cells were then incubated with two different concentrations of nickel nanowires. The cells internalized most wires in less than an hour. The internalization of the lower concentration of nanowires did not lead to significant cell death, however, promisingly, the internalization of the higher concentration resulted in the death of 11 percent of the cancer cells.
Next the researchers exposed the nanowire-containing cells to an alternating magnetic field and noticed a more significant result: the number of cells killed reached 38 percent when the cells, incubated with the higher nanowire concentration, were exposed to a 1 kHz alternating magnetic field.
The researchers suggest that applying the magnetic field triggered vibrations in the nanowires and generated changes in cell functions and structures that eventually led to cell death. Changing the duration of exposure to the magnetic field from 10 to 30 minutes made no relevant difference.
“There have only been a few attempts to study magnetic nanowires for the treatment of cancer cells,” says Kosel. “We are the first ones to use nanowires with only a few tens of nanometers in diameter,” he says.
This method shows promise for future potential medical applications. It allows remote control of nanowires localized in cancer cells, triggering a specific response. Also, since only weak magnetic fields are required to obtain significant results, “they are not harmful to people and can be generated by simple, cheap and compact tools,” explains Maria Contreras, PhD student and first author of the study.
Kosel stresses that many factors still need to be taken into account before this new method can be employed. The team plans to continue improving the efficiency of the method and testing it on various cancer cells.
References
- Contreras, M. F., Sougrat, R. Zaher, A., Ravasi, T. & Kosel., J. Non-chemotoxic induction of cancer cell death using magnetic nanowires. International Journal of Nanomedicine 10, 2141-2153 (2015).| article
You might also like

Electrical Engineering
Seeing smarter with a light-controlled synaptic device

Electrical Engineering
Gallium oxide electronics withstand extreme cold

Computer Science
AI-based hydrogen plant models improve power grid stability

Electrical Engineering
Quantum dot lasers outshine expectations

Bioengineering
Self-aware biosensors boost digital health monitoring

Bioengineering
Smart patch detects allergies before symptoms strike

Computer Science
Green quantum computing takes to the skies

Electrical Engineering



